
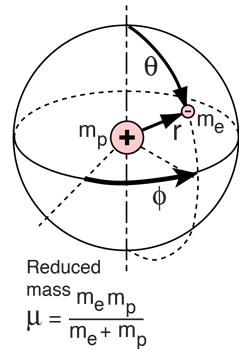
This result is slightly different from that found with Bohr’s theory, which quantizes angular momentum according to the rule L = n, where n = 1, 2, 3. x = r sin θ cos ϕ, y = r sin θ sin ϕ, z = r cos θ. The relationship between spherical and rectangular coordinates is x = r sin θ cos ϕ, y = r sin θ sin ϕ, z = r cos θ. In spherical coordinates, the variable r is the radial coordinate, θ θ is the polar angle (relative to the vertical z-axis), and ϕ ϕ is the azimuthal angle (relative to the x-axis). A spherical coordinate system is shown in Figure 8.3. This suggests that we may solve Schrödinger’s equation more easily if we express it in terms of the spherical coordinates ( r, θ, ϕ ) ( r, θ, ϕ ) instead of rectangular coordinates ( x, y, z ) ( x, y, z ).
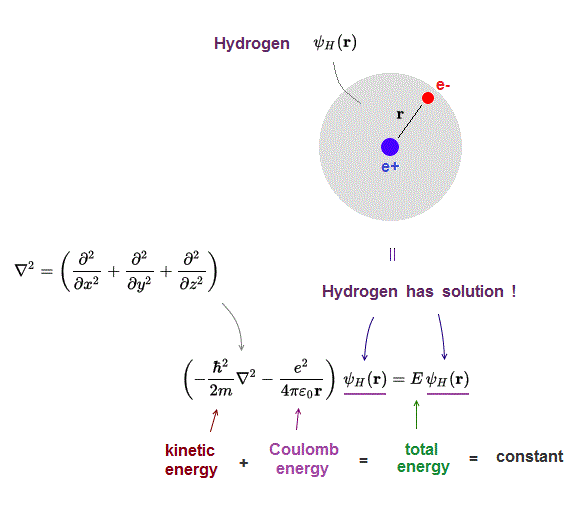
In addition to being time-independent, U( r) is also spherically symmetrical. Recall that the total wave function Ψ ( x, y, z, t ), Ψ ( x, y, z, t ), is the product of the space-dependent wave function ψ = ψ ( x, y, z ) ψ = ψ ( x, y, z ) and the time-dependent wave function φ = φ ( t ) φ = φ ( t ). Where ψ = ψ ( x, y, z ) ψ = ψ ( x, y, z ) is the three-dimensional wave function of the electron, m e m e is the mass of the electron, and E is the total energy of the electron. (This is analogous to the Earth-Sun system, where the Sun moves very little in response to the force exerted on it by Earth.) An explanation of this effect using Newton’s laws is given in Photons and Matter Waves. The proton is approximately 1800 times more massive than the electron, so the proton moves very little in response to the force on the proton by the electron. In Bohr’s model, the electron is pulled around the proton in a perfectly circular orbit by an attractive Coulomb force. The hydrogen atom consists of a single negatively charged electron that moves about a positively charged proton ( Figure 8.2). The hydrogen atom is the simplest atom in nature and, therefore, a good starting point to study atoms and atomic structure. Use quantum numbers to calculate important information about the hydrogen atom.Distinguish between the Bohr and Schrödinger models of the atom.Identify the physical significance of each of the quantum numbers ( n, l, m n, l, m) of the hydrogen atom.Describe the hydrogen atom in terms of wave function, probability density, total energy, and orbital angular momentum.By the end of this section, you will be able to:
